Start with a video by Pediatric Nurse and Doctors and their anecdotal observations, impressions.
Comments
Leave a Reply
Leave a Reply You must Register or Login to post a comment.
Obstetrician, Dr. James Thorp, and Postpartum Nurse, Michelle Gershman, speak out about unprecedented elevation of reproductive and pregnancy complications that have been reported that directly coincide with the Covid-19 vaccine rollout.
#JamesThorpMD #FetalDeathWhistleblower
Canadian Surveillance of COVID-19 in Pregnancy: Epidemiology, Maternal and Infant Outcomes
PLEASE NOTE: Due to changes in COVID-19 testing and reporting across the country we will no longer be updating the map on our website. For the Canadian epidemiology of COVID-19 in pregnancy, we recommend referencing our regularly published reports and manuscripts linked below.
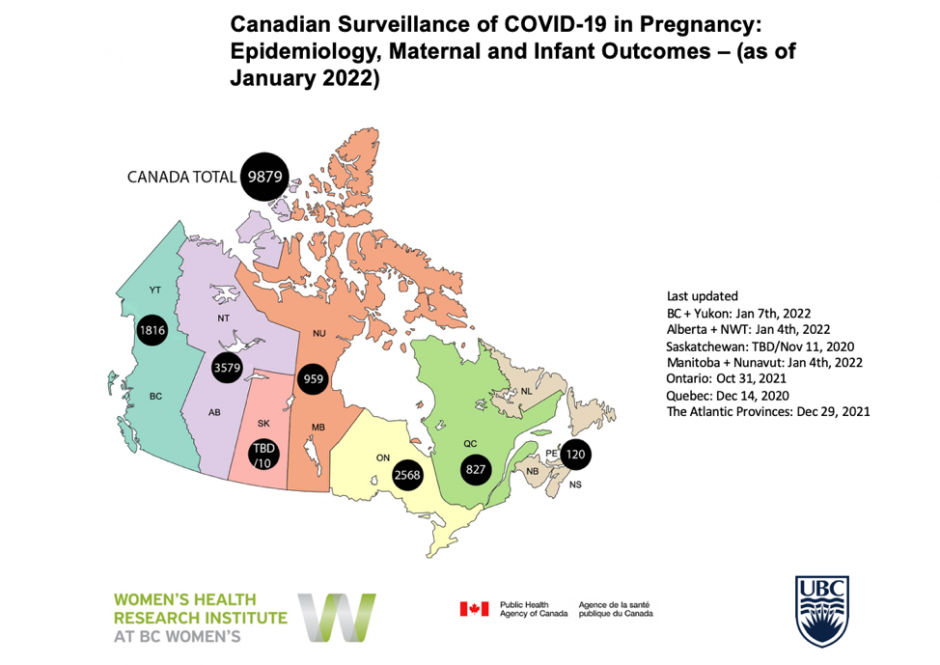
Globally, there is limited data on COVID-19 in pregnancy to inform recommendations for pregnant women and their care providers. This national surveillance project serves to better our understanding of the epidemiology and outcomes associated with COVID-19 in pregnancy and to thereby provide critical data to inform recommendations for pregnant women and their infants. A national approach, with regional leadership across Canada, is in progress with provincial/territorial data collection being combined for a national dataset of cases of COVID-19 in pregnancy. This project also lays the foundation for additional COVID-19 research through the establishment of a biospecimen repository from COVID-19 affected mother-infant pairs.
Coordinating centre lead: Dr. Deborah Money, Professor, Obstetrics & Gynaecology, Medicine, & SPPH Clinician Scientist, Women’s Health Research Institute
For further information please email the CANCOVID-Preg Research Manager Tiffany Reeve at tiffany.reeve@cw.bc.ca Follow @CANCOVIDPreg
Biorepository
As a sub-protocol of the Canadian Surveillance of COVID-19 in Pregnancy: Epidemiology, Maternal and Infant Outcomes, the Rapid Response Maternal-Infant Clinical and Biospecimen Access Platform, or Biorepository Study was developed to better understand the burden of COVID-19 on pregnancy. To do this, we have established a Biorepository of data and samples for future studies related to COVID-19 in pregnant populations.
Recruitment for the Biorepository Study is now closed.
For further information on the bio repository sub-study please reference our study page: Biorepository Sub Study
For further supporting information please reference the sections below
Reports
Protocols
Data Collection Forms
Publications
Supporting Information

Reproductive Infectious Diseases Program | Department of Obstetrics & Gynaecology
Women’s Health Research Institute
4500 Oak Street
Vancouver, BCCanadaV6H 3N1
Email rid.program@ubc.ca
Effects of Vaccination on Sperm
Multicenter Study Andrology
. 2022 Sep;10(6):1016-1022.
doi: 10.1111/andr.13209. Epub 2022 Jun 27.
Covid-19 vaccination BNT162b2 temporarily impairs semen concentration and total motile count among semen donors
Itai Gat 1 2 3 , Alon Kedem 2 3 4 , Michal Dviri 5 , Ana Umanski 1 , Matan Levi 4 , Ariel Hourvitz 2 3 , Micha Baum 3 6
Affiliations
Results: Repetitive measurements revealed -15.4% sperm concentration decrease on T2 (CI -25.5%-3.9%, p = 0.01) leading to total motile count 22.1% reduction (CI -35% – -6.6%, p = 0.007) compared to T0. Similarly, analysis of first semen sample only and samples’ mean per donor resulted in concentration and total motile count (TMC) reductions on T2 compared to T0 – median decline of 12 million/ml and 31.2 million motile spermatozoa, respectively (p = 0.02 and 0.002 respectively) on first sample evaluation and median decline of 9.5 × 106 and 27.3 million motile spermatozoa (p = 0.004 and 0.003, respectively) on samples’ mean examination. T3 evaluation demonstrated overall recovery without. Semen volume and sperm motility were not impaired.
Research Letter
June 17, 2021
Sperm Parameters Before and After COVID-19 mRNA Vaccination
Daniel C. Gonzalez, BS1; Daniel E. Nassau, MD1; Kajal Khodamoradi, PhD1; et alEmad Ibrahim, MD1; Ruben Blachman-Braun, MD1; Jesse Ory, MD1; Ranjith Ramasamy, MD1
Author AffiliationsArticle Information
JAMA. 2021;326(3):273-274. doi:10.1001/jama.2021.9976
Between December 17, 2020, and January 12, 2021, 45 men volunteered (median age, 28 years [IQR, 25-31]); follow-up samples were obtained at a median of 75 days (IQR, 70-86) after the second dose. The study ended on April 24, 2021. Baseline samples were obtained after a median abstinence period of 2.8 days (IQR, 2-3) and follow-up samples after a median of 3 days (IQR, 3-4). Of the 45 men, 21 (46.7%) received BNT162b2 and 24 (53.3%) received mRNA-1273. Baseline median sperm concentration and TMSC were 26 million/mL (IQR, 19.5-34) and 36 million (IQR, 18-51), respectively. After the second vaccine dose, the median sperm concentration significantly increased to 30 million/mL (IQR, 21.5-40.5; P = .02) and the median TMSC to 44 million (IQR, 27.5-98; P = .001). Semen volume and sperm motility also significantly increased (Table).
- https://www.bornontario.ca/en/pso/prenatal-screening-options/covid-19-and-pregnancy.aspx
- https://www.omama.com/en/pre-pregnancy/coronavirus-disease–covid-19-.asp
- https://www.canada.ca/en/public-health/services/immunization-vaccines/vaccination-pregnancy-covid-19.html
- https://www.cdc.gov/vaccines/pregnancy/index.html
- https://www.cdc.gov/coronavirus/2019-ncov/vaccines/recommendations/pregnancy.html
- https://www.hopkinsmedicine.org/health/conditions-and-diseases/coronavirus/covid19-vaccine-hesitancy-12-things-you-need-to-know
- A good decision aid produced in Ontario is located here: https://www.pcmch.on.ca/wp-content/uploads/2021/10/PCMCH-COVID-19-Vaccine-Pregnancy-Information-Sheet-2021Oct25_v4.pdf
References:
1 Money et al. Canadian Surveillance of COVID-19 in Pregnancy: Epidemiology, Maternal and Infant Outcomes (2021). https://med-fom-ridprogram.sites.olt.ubc.ca/files/2021/10/CANCOVID_Preg-report-4-19oct2021.pdf
2 Vousden et al. Impact of SARS-CoV-2 variant on the severity of maternal infection and perinatal outcomes: Data from the UK Obstetric Surveillance System national cohort (2021). https://www.medrxiv.org/content/10.1101/2021.07.22.21261000v1
3 Allotey et al. Clinical manifestations, risk factors, and maternal and perinatal outcomes of coronavirus disease 2019 in pregnancy: living systematic review and meta-analysis. (2019) BMJ Sep 1:370. https://pubmed.ncbi.nlm.nih.gov/32873575/
4 Jering et al. Clinical Characteristics and Outcomes of Hospitalized Women Giving Birth With and Without COVID-19. (2021) JAMA Intern Med May 1;181(5):714-717. https://pubmed.ncbi.nlm.nih.gov/?sort=date&term=Claggett+BL&cauthor_id=33449067
Contact Us
BORN Ontario
Centre for Practice-Changing Research Building (CPCR)
401 Smyth Road | Ottawa, ON K1H 8L1
T. 613-737-7600 x6022
E-Mail this contact
Website
Map this Location